PROGRAM
COMPASS TNBC
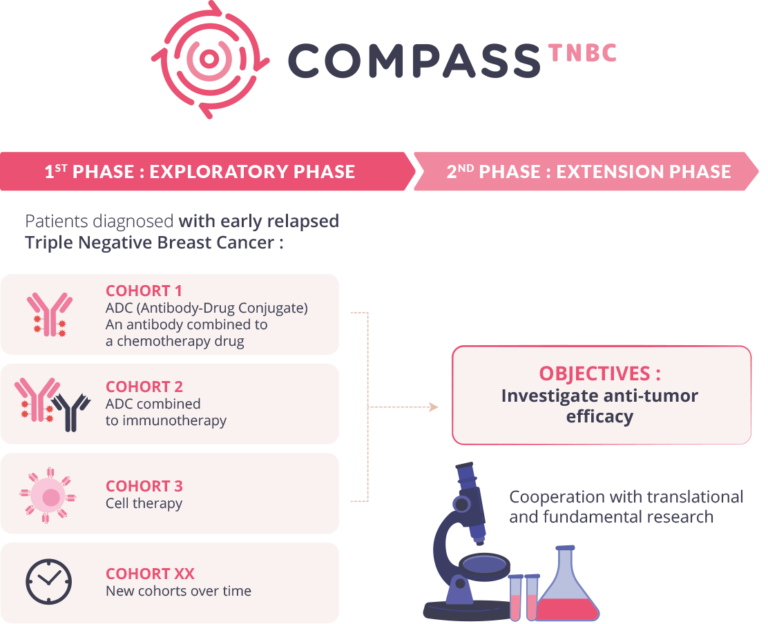
The COMPASS-TNBC (Choice Of the Most Active Strategies for Short term recurring triple negative breast cancer) program consists of several phase I/II clinical trials to evaluate the effectiveness of innovative therapeutic strategies in patients with early-relapsed metastatic triple negative breast cancer.
Phase I/II trials allow for a preliminary assessment of efficacy and the investigation of innovative combination drug therapies. The COMPASS-TNBC program comprises a series of rapidly enrolling Phase I/II trials conducted in two stages. The first stage is an exploratory phase involving a limited number of patients per treatment module. If an activity signal is observed, an extension phase will be initiated, enrolling several dozen or even a hundred patients, to confirm preliminary antitumour activity and assess treatment tolerability.
The COMPASS-TNBC program is based on a partnership between Gustave Roussy and several laboratories with the involvement of multiple translational research teams, to deepen our knowledge of tumour evolution and mechanisms of treatment resistance.
The treatments investigated in this program are selected by a multidisciplinary committee comprising medical oncologists specialized in the treatment of triple-negative breast cancer, immunologists, fundamental and translational research specialists.
JOINING THE PROGRAM
HOW TO JOIN COMPASS-TNBC
Patients or their healthcare providers can reach out to our team by filling out a designated form on this website.
Our team will promptly respond within a week. If the patient meets the inclusion criteria, an initial online consultation will be conducted to provide further information about the study, the type of treatment and potential side effects and to assess the patient’s eligibility for the study.